
| |
| Names | |
|---|---|
|
IUPAC name
2,6,6-trimethylcyclohexene-1-carbaldehyde
| |
| Other names
Beta-cyclocitral, B-cyclocitral
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 2042086 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.439 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C10H16O | |
| Molar mass | 152.237 g·mol−1 |
| Boiling point | 62–63 °C (144–145 °F; 335–336 K) |
| 86.14 mg/L | |
| Hazards | |
| GHS labelling: [1] | |

| |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P317, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Cyclocitral (beta-cyclocitral) is an apocarotenoid derived from the C7 oxidation of β-carotene. This apocarotenoid has revived interest due to its roles in plant development. β-cyclocitral has been found endogenously in a variety of organisms including plants, cyanobacteria, fungi and animals. [2] β-Cyclocitral is a volatile compound that contributes to the aroma of various fruits, vegetables and ornamental plants. [3] In plants, β-cyclocitral was found to be an important regulator in root development. [4]
Application
β-Cyclocitral is used as an analytical standard for the determination of volatile organic compounds in saffron due to its analog structure to safranal.
Because β-cyclocitral is associated with cyanobacteria death, it is an analyte that can be tracked in bodies of water to monitor cyanobacteria blooms. [5]
It has also been found to promote the growth of roots in rice, prompting its consideration as a potential agricultural tool. [6]
Biosynthesis
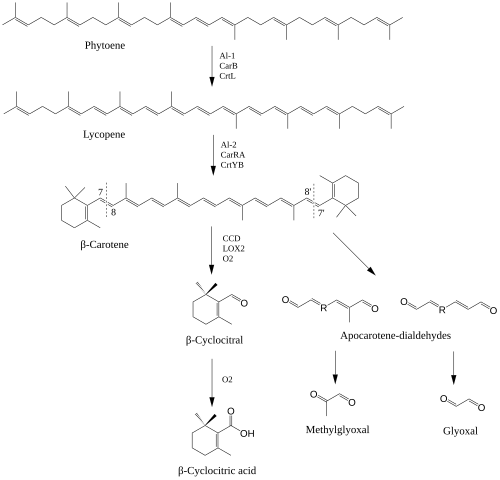
The biosynthesis of β-cyclocitral relies on the formation of β-carotene through the isoprenoid biosynthetic pathway underpinning carotenoid formation. Similar to other apocarotenoids, the formation of β-cyclocitral can occur via the enzymatic and non-enzymatic oxidative cleavage of double bonds in β-carotene. [7] For β-cyclocitral to form, the cleavage of C7-C8 double bonds are needed. While no enzyme has been identified to have high specificity for the production of β-cyclocitral, a carotenoid cleavage dioxygenase (CCD4) has been identified as being capable of cleaving β-carotene at the needed position. [8] 13-lipoxygenase (LOX2) has also been identified to cleave β-carotene at the C7 position. [9] β-cyclocitral can also be formed from the direct oxidation of β-carotene by reactive oxygen species, especially singlet oxygen (1O2). In plants, 1O2 is mainly produced from excited chlorophylls in the reaction center of PSII where β-carotene serves to quench the reactive oxygen species. [10]
References
- ^ "beta-Cyclocitral". pubchem.ncbi.nlm.nih.gov.
- ^ Havaux, Michel (October 2020). "β-Cyclocitral and derivatives: Emerging molecular signals serving multiple biological functions". Plant Physiology and Biochemistry. 155: 35–41. doi: 10.1016/j.plaphy.2020.07.032. ISSN 0981-9428. PMID 32738580. S2CID 220925143.
- ^ Condurso, Concetta (October 2016). "Bioactive volatiles in Sicilian (South Italy) saffron: safranal and its related compounds". Journal of Essential Oil Research. 29 (3): 221–227. doi: 10.1080/10412905.2016.1244115. S2CID 100505185.
- ^ Dickinson, Alexandra (May 2019). "β-Cyclocitral is a conserved root growth regulator". Proceedings of the National Academy of Sciences. 116 (21): 10563–10567. Bibcode: 2019PNAS..11610563D. doi: 10.1073/pnas.1821445116. PMC 6534974. PMID 31068462.
- ^ Huang, Heyong (2018). "Distributions of four taste and odor compounds in the sediment and overlying water at different ecology environment in Taihu Lake". Scientific Reports. 8 (8): 6179. Bibcode: 2018NatSR...8.6179H. doi: 10.1038/s41598-018-24564-z. PMC 5906450. PMID 29670292.
- ^ Keeley, Jim. "A Plant Hormone that Speeds Root Growth Could Be a New Agricultural Tool". Howard Hughhes Medical Institute. Retrieved 6 June 2023.
- ^ Havaux, Michel (2020). "β-Cyclocitral and derivatives: Emerging molecular signals serving multiple biological functions". Plant Physiology and Biochemistry. 155: 35–41. doi: 10.1016/j.plaphy.2020.07.032. PMID 32738580. S2CID 220925143.
- ^ Maria, Rodrigo (2013). "A novel carotenoid cleavage activity involved in the biosynthesis of Citrus fruit-specific apocarotenoid pigments". Journal of Experimental Botany. 64 (14): 4461–4478. doi: 10.1093/jxb/ert260. PMC 3808326. PMID 24006419.
- ^ Gao, Lei (2019). "The tomato pan-genome uncovers new genes and a rare allele regulating fruit flavor". Nature Genetics. 51 (6): 1044–1051. doi: 10.1038/s41588-019-0410-2. PMID 31086351. S2CID 256819279.
- ^ Triantaphylidès, Christian (2009). "Singlet oxygen in plants: production, detoxification and signaling". Trends in Plant Science. 14 (4): 219–228. doi: 10.1016/j.tplants.2009.01.008. PMID 19303348.

| |
| Names | |
|---|---|
|
IUPAC name
2,6,6-trimethylcyclohexene-1-carbaldehyde
| |
| Other names
Beta-cyclocitral, B-cyclocitral
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 2042086 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.439 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C10H16O | |
| Molar mass | 152.237 g·mol−1 |
| Boiling point | 62–63 °C (144–145 °F; 335–336 K) |
| 86.14 mg/L | |
| Hazards | |
| GHS labelling: [1] | |

| |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P317, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Cyclocitral (beta-cyclocitral) is an apocarotenoid derived from the C7 oxidation of β-carotene. This apocarotenoid has revived interest due to its roles in plant development. β-cyclocitral has been found endogenously in a variety of organisms including plants, cyanobacteria, fungi and animals. [2] β-Cyclocitral is a volatile compound that contributes to the aroma of various fruits, vegetables and ornamental plants. [3] In plants, β-cyclocitral was found to be an important regulator in root development. [4]
Application
β-Cyclocitral is used as an analytical standard for the determination of volatile organic compounds in saffron due to its analog structure to safranal.
Because β-cyclocitral is associated with cyanobacteria death, it is an analyte that can be tracked in bodies of water to monitor cyanobacteria blooms. [5]
It has also been found to promote the growth of roots in rice, prompting its consideration as a potential agricultural tool. [6]
Biosynthesis
The biosynthesis of β-cyclocitral relies on the formation of β-carotene through the isoprenoid biosynthetic pathway underpinning carotenoid formation. Similar to other apocarotenoids, the formation of β-cyclocitral can occur via the enzymatic and non-enzymatic oxidative cleavage of double bonds in β-carotene. [7] For β-cyclocitral to form, the cleavage of C7-C8 double bonds are needed. While no enzyme has been identified to have high specificity for the production of β-cyclocitral, a carotenoid cleavage dioxygenase (CCD4) has been identified as being capable of cleaving β-carotene at the needed position. [8] 13-lipoxygenase (LOX2) has also been identified to cleave β-carotene at the C7 position. [9] β-cyclocitral can also be formed from the direct oxidation of β-carotene by reactive oxygen species, especially singlet oxygen (1O2). In plants, 1O2 is mainly produced from excited chlorophylls in the reaction center of PSII where β-carotene serves to quench the reactive oxygen species. [10]

References
- ^ "beta-Cyclocitral". pubchem.ncbi.nlm.nih.gov.
- ^ Havaux, Michel (October 2020). "β-Cyclocitral and derivatives: Emerging molecular signals serving multiple biological functions". Plant Physiology and Biochemistry. 155: 35–41. doi: 10.1016/j.plaphy.2020.07.032. ISSN 0981-9428. PMID 32738580. S2CID 220925143.
- ^ Condurso, Concetta (October 2016). "Bioactive volatiles in Sicilian (South Italy) saffron: safranal and its related compounds". Journal of Essential Oil Research. 29 (3): 221–227. doi: 10.1080/10412905.2016.1244115. S2CID 100505185.
- ^ Dickinson, Alexandra (May 2019). "β-Cyclocitral is a conserved root growth regulator". Proceedings of the National Academy of Sciences. 116 (21): 10563–10567. Bibcode: 2019PNAS..11610563D. doi: 10.1073/pnas.1821445116. PMC 6534974. PMID 31068462.
- ^ Huang, Heyong (2018). "Distributions of four taste and odor compounds in the sediment and overlying water at different ecology environment in Taihu Lake". Scientific Reports. 8 (8): 6179. Bibcode: 2018NatSR...8.6179H. doi: 10.1038/s41598-018-24564-z. PMC 5906450. PMID 29670292.
- ^ Keeley, Jim. "A Plant Hormone that Speeds Root Growth Could Be a New Agricultural Tool". Howard Hughhes Medical Institute. Retrieved 6 June 2023.
- ^ Havaux, Michel (2020). "β-Cyclocitral and derivatives: Emerging molecular signals serving multiple biological functions". Plant Physiology and Biochemistry. 155: 35–41. doi: 10.1016/j.plaphy.2020.07.032. PMID 32738580. S2CID 220925143.
- ^ Maria, Rodrigo (2013). "A novel carotenoid cleavage activity involved in the biosynthesis of Citrus fruit-specific apocarotenoid pigments". Journal of Experimental Botany. 64 (14): 4461–4478. doi: 10.1093/jxb/ert260. PMC 3808326. PMID 24006419.
- ^ Gao, Lei (2019). "The tomato pan-genome uncovers new genes and a rare allele regulating fruit flavor". Nature Genetics. 51 (6): 1044–1051. doi: 10.1038/s41588-019-0410-2. PMID 31086351. S2CID 256819279.
- ^ Triantaphylidès, Christian (2009). "Singlet oxygen in plants: production, detoxification and signaling". Trends in Plant Science. 14 (4): 219–228. doi: 10.1016/j.tplants.2009.01.008. PMID 19303348.