This article may be too technical for most readers to understand. (May 2023) |

| |||

| |||
| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
2-(2-nitrophenyl)acetic acid
[2]
| |||
| Other names
Benzeneacetic acid, 2-nitro-
[1]
o-Nitrophenylacetic acid 2-nitrophenylacetic acid (ortho-Nitrophenyl)acetic acid acetic acid, (o-nitrophenyl) 2-(o-nitrophenyl)acetic acid | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.021.026 | ||
| EC Number |
| ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C8H7NO4 | |||
| Molar mass | 181.15 g/mol | ||
| Appearance | Yellow to Pale Brown Crystalline Powder | ||
| Density | 1.4 g/cm3 [3] | ||
| Boiling point | 141 °C (286 °F; 414 K) | ||
| 0.1417% (20 °C) [4] | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| GHS labelling: | |||


| |||
| Warning | |||
| H315, H319, H335, H341 | |||
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P317, P319, P321, P332, P362+P364, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | MSDS | ||
| Related compounds | |||
Related derivatives;
related aromatic compounds |
phenylacetic acid,
4-nitrophenylacetic acid; 4-nitrophenol, 2-nitrodiphenylamine | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Nitrophenylacetic acid is an organic compound used in organic synthesis that has also been used as an herbicide. It is a derivative of phenylacetic acid, containing a phenyl functional group, a carboxylic acid functional group, and a nitro functional group. It is an important reagent for many organic reactions, especially for the formation of heterocycles.
Synthesis
This compound may be prepared by the
nitration of
phenylacetic acid.
[5]

Applications
In organic synthesis, 2-nitrophenylacetic acid can be used as a
protecting group for primary
alcohols. The alcohol is
esterified with 2-nitrophenylacetic acid, proceeding through the
acid chloride or
acid anhydride. The acid itself can also protect the alcohol through the
Mitsunobu reaction: reacting the alcohol and the acid with
diethyl azidocarboxylate and
triphenylphosphine in
dichloromethane. The protecting group is selectively removed using
zinc and
ammonium chloride, and is compatible with other existing alcohol protecting groups.
[6]

In addition, 2-nitrophenylacetic acid is a
precursor for many
heterocycles. Complete
reduction of 2-nitrophenylacetic acid yields
anilines, which quickly cyclize to form
lactams.
[7]
[8] Partial reductive
cyclization of the acids using weaker
reducing agents forms
hydroxamic acids.
[8]
Both of these processes are useful in the synthesis of many
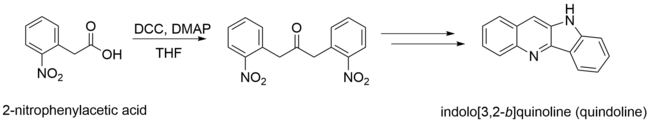
biologically active molecules. 2-nitrophenylacetic acid is a precursor of
quindoline, which although it does not have many practical applications on its own, quindoline derivatives and modifications can be treated as enzyme inhibitors and anticancer agents.
[9]
Derivatives of 2-nitrophenylacetic acids are useful in total synthesis for their ability to form heterocycles. 2-nitrophenylacetic acid is a precursor to (−)-phaitanthrin D, a clinically useful molecule originally isolated from the Phaius mishmensis orchid. [10] The carboxylic acid on the 2-nitrophenylacetic acid is first protected using menthol, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCl), hydroxybenzotriazole(HOBt) and N,N-iisopropylethylamine(DIPEA). A pattern of reducing the nitro group to an amino group and subsequently forming amides by the addition to carboxylic acids (namely nitrobenzoic acid) occurs. Reductive cyclization of the subsequent product using hexamethyldisilazane, zinc chloride and dimethylformamide forms the disubstituted heterocycle present in the (−)-phaitantrin D molecule.

Outside of organic synthesis, 2-nitrophenylacetic acid has been used as an
herbicide, as it displays selective herbicidal properties.
[11] It has also been used as an
internal standard for measurement of salicylamide-O-acetic acid (an
anti-asthma drug) using
high performance liquid chromatography.
[12]
References
- ^ "Benzeneacetic acid, 2-nitro-". NIST Chemistry WebBook, SRD 69.
- ^ "2-Nitrophenylacetic acid (compound)". PubChem. National Library of Medicine. Retrieved 14 May 2023.
- ^ "AJ1130000". ChemSpider. Retrieved 14 May 2023.
- ^ Zhou, Yanyan; Wu, Jiaxin; Wang, Jian; Zhao, Hongkun (13 August 2020). "Equilibrium Solubility and Dissolution Property Analysis of 2-Nitrophenylacetic Acid in 13 Pure Solvents at Elevated Temperatures". Journal of Chemical & Engineering Data. 65 (8): 4157–4165. doi: 10.1021/acs.jced.0c00543. S2CID 225475559.
- ^ Sohail, Muhammad; Raza, Abdul Rauf (February 2012). "A Novel One Pot Synthesis of o-Nitrophenylacetic Acid and Unexpected p-Nitrobenzoic Acid by HNO3-Mediated CH2 Extrusion Reaction of Phenylacetic Acid". Chinese Journal of Chemistry. 30 (2): 353–356. doi: 10.1002/cjoc.201180458.
- ^ Daragics, Katalin; Fügedi, Péter (7 May 2010). "(2-Nitrophenyl)acetyl: A New, Selectively Removable Hydroxyl Protecting Group". Organic Letters. 12 (9): 2076–2079. doi: 10.1021/ol100562f. PMID 20361745.
- ^ Wright, William B.; Collins, Kenneth H. (January 1956). "Cyclic Hydroxamic Acids Derived from Indole". Journal of the American Chemical Society. 78 (1): 221–224. doi: 10.1021/ja01582a061. ISSN 0002-7863.
- ^ a b Ichire, Ogar; Jans, Petra; Parfenov, Galina; Dounay, Amy B. (8 February 2017). "A safe and selective method for reduction of 2-nitrophenylacetic acid systems to N-aryl hydroxamic acids using continuous flow hydrogenation". Tetrahedron Letters. 58 (6): 582–585. doi: 10.1016/j.tetlet.2017.01.008. ISSN 0040-4039.
- ^ Singer, Jamie M.; Barr, Bridget M.; Coughenour, Linda L.; Gregory, Tracy F.; Walters, Michael A. (15 October 2005). "8-Substituted 3,4-dihydroquinolinones as a novel scaffold for atypical antipsychotic activity". Bioorganic & Medicinal Chemistry Letters. 15 (20): 4560–4563. doi: 10.1016/j.bmcl.2005.06.097. ISSN 0960-894X. PMID 16087333.
- ^ Vaidya, Sagar; Argade, Narshinha (17 May 2016). "Synthesis of (–)-Phaitanthrin D and (+)-Dihydropyrroloindoloquinazolinone". Synthesis. 48 (17): 2896–2903. doi: 10.1055/s-0035-1562098. ISSN 0039-7881. S2CID 99029333.
- ^ Back, Gayle E.; Dahle, Norman A. (6 July 1971). "NITROPHENYLACETIC ACID DERVATIVES" (PDF). United States Patent Office.
- ^ Schulz, H. -U.; Kraas, E. (1 January 1985). "Determination of the theophylline solubilizer salicylamide-O-acetic acid in serum and urine using high-performance liquid chromatography". Journal of Pharmaceutical and Biomedical Analysis. 3 (5): 469–475. doi: 10.1016/0731-7085(85)80062-5. ISSN 0731-7085. PMID 16867660.
This article may be too technical for most readers to understand. (May 2023) |

| |||

| |||
| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
2-(2-nitrophenyl)acetic acid
[2]
| |||
| Other names
Benzeneacetic acid, 2-nitro-
[1]
o-Nitrophenylacetic acid 2-nitrophenylacetic acid (ortho-Nitrophenyl)acetic acid acetic acid, (o-nitrophenyl) 2-(o-nitrophenyl)acetic acid | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.021.026 | ||
| EC Number |
| ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C8H7NO4 | |||
| Molar mass | 181.15 g/mol | ||
| Appearance | Yellow to Pale Brown Crystalline Powder | ||
| Density | 1.4 g/cm3 [3] | ||
| Boiling point | 141 °C (286 °F; 414 K) | ||
| 0.1417% (20 °C) [4] | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| GHS labelling: | |||


| |||
| Warning | |||
| H315, H319, H335, H341 | |||
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P317, P319, P321, P332, P362+P364, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | MSDS | ||
| Related compounds | |||
Related derivatives;
related aromatic compounds |
phenylacetic acid,
4-nitrophenylacetic acid; 4-nitrophenol, 2-nitrodiphenylamine | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Nitrophenylacetic acid is an organic compound used in organic synthesis that has also been used as an herbicide. It is a derivative of phenylacetic acid, containing a phenyl functional group, a carboxylic acid functional group, and a nitro functional group. It is an important reagent for many organic reactions, especially for the formation of heterocycles.
Synthesis
This compound may be prepared by the
nitration of
phenylacetic acid.
[5]

Applications
In organic synthesis, 2-nitrophenylacetic acid can be used as a
protecting group for primary
alcohols. The alcohol is
esterified with 2-nitrophenylacetic acid, proceeding through the
acid chloride or
acid anhydride. The acid itself can also protect the alcohol through the
Mitsunobu reaction: reacting the alcohol and the acid with
diethyl azidocarboxylate and
triphenylphosphine in
dichloromethane. The protecting group is selectively removed using
zinc and
ammonium chloride, and is compatible with other existing alcohol protecting groups.
[6]

In addition, 2-nitrophenylacetic acid is a
precursor for many
heterocycles. Complete
reduction of 2-nitrophenylacetic acid yields
anilines, which quickly cyclize to form
lactams.
[7]
[8] Partial reductive
cyclization of the acids using weaker
reducing agents forms
hydroxamic acids.
[8]
Both of these processes are useful in the synthesis of many
biologically active molecules. 2-nitrophenylacetic acid is a precursor of
quindoline, which although it does not have many practical applications on its own, quindoline derivatives and modifications can be treated as enzyme inhibitors and anticancer agents.
[9]

Derivatives of 2-nitrophenylacetic acids are useful in total synthesis for their ability to form heterocycles. 2-nitrophenylacetic acid is a precursor to (−)-phaitanthrin D, a clinically useful molecule originally isolated from the Phaius mishmensis orchid. [10] The carboxylic acid on the 2-nitrophenylacetic acid is first protected using menthol, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCl), hydroxybenzotriazole(HOBt) and N,N-iisopropylethylamine(DIPEA). A pattern of reducing the nitro group to an amino group and subsequently forming amides by the addition to carboxylic acids (namely nitrobenzoic acid) occurs. Reductive cyclization of the subsequent product using hexamethyldisilazane, zinc chloride and dimethylformamide forms the disubstituted heterocycle present in the (−)-phaitantrin D molecule.

Outside of organic synthesis, 2-nitrophenylacetic acid has been used as an
herbicide, as it displays selective herbicidal properties.
[11] It has also been used as an
internal standard for measurement of salicylamide-O-acetic acid (an
anti-asthma drug) using
high performance liquid chromatography.
[12]
References
- ^ "Benzeneacetic acid, 2-nitro-". NIST Chemistry WebBook, SRD 69.
- ^ "2-Nitrophenylacetic acid (compound)". PubChem. National Library of Medicine. Retrieved 14 May 2023.
- ^ "AJ1130000". ChemSpider. Retrieved 14 May 2023.
- ^ Zhou, Yanyan; Wu, Jiaxin; Wang, Jian; Zhao, Hongkun (13 August 2020). "Equilibrium Solubility and Dissolution Property Analysis of 2-Nitrophenylacetic Acid in 13 Pure Solvents at Elevated Temperatures". Journal of Chemical & Engineering Data. 65 (8): 4157–4165. doi: 10.1021/acs.jced.0c00543. S2CID 225475559.
- ^ Sohail, Muhammad; Raza, Abdul Rauf (February 2012). "A Novel One Pot Synthesis of o-Nitrophenylacetic Acid and Unexpected p-Nitrobenzoic Acid by HNO3-Mediated CH2 Extrusion Reaction of Phenylacetic Acid". Chinese Journal of Chemistry. 30 (2): 353–356. doi: 10.1002/cjoc.201180458.
- ^ Daragics, Katalin; Fügedi, Péter (7 May 2010). "(2-Nitrophenyl)acetyl: A New, Selectively Removable Hydroxyl Protecting Group". Organic Letters. 12 (9): 2076–2079. doi: 10.1021/ol100562f. PMID 20361745.
- ^ Wright, William B.; Collins, Kenneth H. (January 1956). "Cyclic Hydroxamic Acids Derived from Indole". Journal of the American Chemical Society. 78 (1): 221–224. doi: 10.1021/ja01582a061. ISSN 0002-7863.
- ^ a b Ichire, Ogar; Jans, Petra; Parfenov, Galina; Dounay, Amy B. (8 February 2017). "A safe and selective method for reduction of 2-nitrophenylacetic acid systems to N-aryl hydroxamic acids using continuous flow hydrogenation". Tetrahedron Letters. 58 (6): 582–585. doi: 10.1016/j.tetlet.2017.01.008. ISSN 0040-4039.
- ^ Singer, Jamie M.; Barr, Bridget M.; Coughenour, Linda L.; Gregory, Tracy F.; Walters, Michael A. (15 October 2005). "8-Substituted 3,4-dihydroquinolinones as a novel scaffold for atypical antipsychotic activity". Bioorganic & Medicinal Chemistry Letters. 15 (20): 4560–4563. doi: 10.1016/j.bmcl.2005.06.097. ISSN 0960-894X. PMID 16087333.
- ^ Vaidya, Sagar; Argade, Narshinha (17 May 2016). "Synthesis of (–)-Phaitanthrin D and (+)-Dihydropyrroloindoloquinazolinone". Synthesis. 48 (17): 2896–2903. doi: 10.1055/s-0035-1562098. ISSN 0039-7881. S2CID 99029333.
- ^ Back, Gayle E.; Dahle, Norman A. (6 July 1971). "NITROPHENYLACETIC ACID DERVATIVES" (PDF). United States Patent Office.
- ^ Schulz, H. -U.; Kraas, E. (1 January 1985). "Determination of the theophylline solubilizer salicylamide-O-acetic acid in serum and urine using high-performance liquid chromatography". Journal of Pharmaceutical and Biomedical Analysis. 3 (5): 469–475. doi: 10.1016/0731-7085(85)80062-5. ISSN 0731-7085. PMID 16867660.



